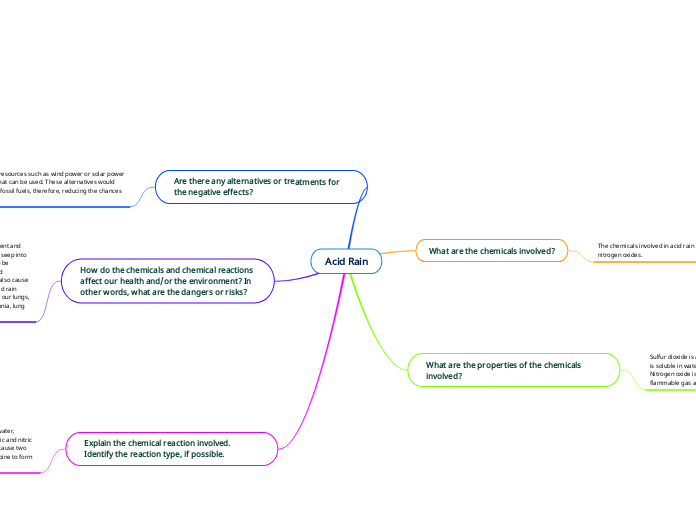
The chemicals involved in acid rain are sulfur dioxide and nitrogen oxides.
Sulfur dioxide is a colourless chemical, with a sharp odour and is soluble in water.
Nitrogen oxide is a colourless chemical with a slight odour, is a flammable gas and is soluble in water.
Using renewable resources such as wind power or solar power is an alternative that can be used. These alternatives would reduce the use of fossil fuels, therefore, reducing the chances of acid rain.
The dangers of acid rain apply to both the environment and our health. Acid rain is a very toxic substance, it can seep into the ground and dissolve nutrients that trees need to be healthy. It can also destroy aquatic life when the acid substance is mixed in the freshwater. Acid rain can also cause corrosion in steel infrastructure such as bridges. Acid rain affects our health because small particles can enter our lungs, therefore, causing health problems such as pneumonia, lung damage and respiratory problems.
The sulfur dioxide and nitrogen oxides react with water, oxygen and various other chemicals to form sulfuric and nitric acids. The reaction type is a synthesis reaction because two chemicals (sulfur dioxide and nitrogen oxide) combine to form one substance (acid rain).