The most abundant biomolecules on earthMainly contain carbon, hydrogen and oxygen atoms
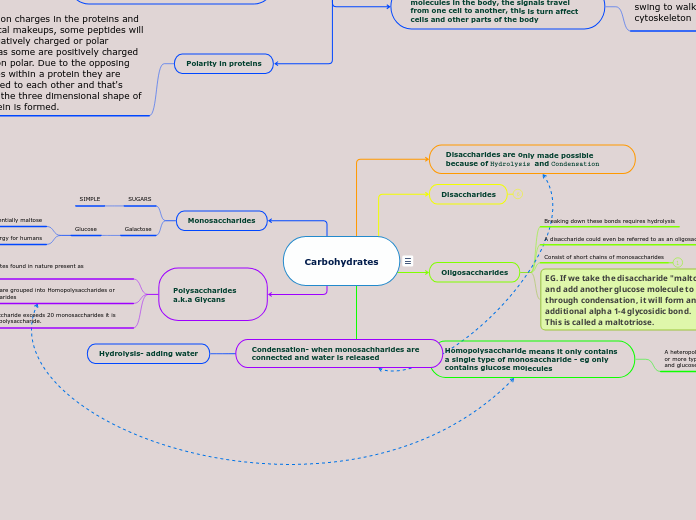
Breaking down these bonds requires hydrolysis
A disaccharide could even be referred to as an oligosaccharide
Consist of short chains of monosaccharides
EG. If we take the disaccharide "maltose" and add another glucose molecule to it, through condensation, it will form an additional alpha 1-4 glycosidic bond.
This is called a maltotriose.
A heteropolysaccharide means the polysaccharide contains two or more types of monosaccharide- eg a long chain of fructose and glucose molecules
SUGARS
SIMPLE
Galactose
Glucose
Two glucose mixed together is essentially maltose
Main source of energy for humans
Most carbohydrates found in nature present as polysaccharides
Polysaccharides are grouped into Homopolysaccharides or Heteropolysaccharides
Once an oligosaccharide exceeds 20 monosaccharides it is referred to as a polysaccharide.
Amino acids are what make up proteins, they're the building blocks.
Every amino acid has a carbon with a hydrogen attached to it, this is in the middle of the carboxyl and amino group and it is called the alpha carbon
Right side has a carboxyl group
Every amino acid looks very similar with these three parts, what distinguishes them is what comes off the alpha carbon called the R groups
The left side has nitrogen attached to two hydrogen makes an amino group
Proteins are flexible and can bend and swing to walk across the cells cytoskeleton
When a polypeptide folds itself, its called a protein and each protein has different chemical properties
Based on charges in the proteins and chemical makeups, some peptides will be negatively charged or polar whereas some are positively charged and non polar. Due to the opposing charges within a protein they are attracted to each other and that's where the three dimensional shape of a protein is formed.
If a protein is non polar it is hydrophobic if it is polar on the other hand its hydrophilic